Room: 3'102 / 3'125
Phone: +49 (0) 30 2093 82653
E-Mail: kristin.klauehu-berlin.de
Dr. Kristin Klaue
Development of One-Photon NIR Photoswitches
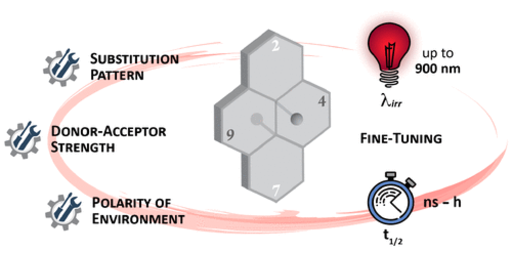
Incorporation of molecular switches in complex molecular systems allows dynamic alteration of their intrinsic properties and function with light in a remote and non-invasive fashion. For most of these applications, visible or near-infrared (NIR) light is more suitable than UV light due to its enhanced penetration depth and reduced radiation damage. However, the isomerization of photoswitches usually requires high-energy photons in at least one direction, which limits their utilization especially in biological applications. We develop tunable direct one-photon NIR photoswitches based on negative photochromic dihydropyrenes carrying opposing strong donor-acceptor substituents either along the long axis of the molecule or across it. This allows large temporary changes of their properties upon visible or NIR light-induced photoisomerization.
2024
Diaryltriazolium Photoswitch: Reaching a Millisecond Cycloreversion with High Stability and NIR Absorption
D. Kolarski*, P. Steinbach, C. Bannwarth*, K. Klaue and S. Hecht*
Angew. Chem. Int. Ed. 2024, 61, e202318015
2023
DiI-CT—A bimodal neural tracer for X-ray and fluorescence imaging
B. Gerhardt, K. Klaue, L. Eigen, J. Schwarz, S. Hecht* and Michael Brecht*
Cell Report Methods 2023, 3, 100486

2021
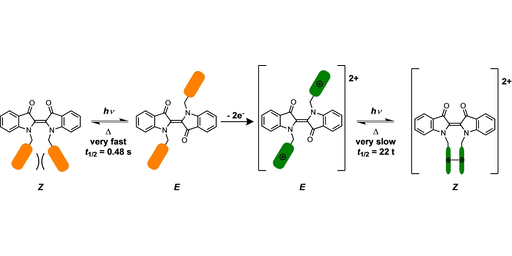
Stabilizing Indigo Z-isomer via Intramolecular Associations of Redox-Active Appendages
L. Broløs, K. Klaue, J. Bendix, L. Grubert, S. Hecht*, M. Brøndsted Nielsen*
2020
Donor-Acceptor Dihydropyrenes Switchable with Near-Infrared Light
K. Klaue, W. Han, P. Liesfeld, F. Berger, Y. Garmshausen, S. Hecht*
2018
Taking Photochromism Beyond Visible: Direct One-Photon NIR Photoswitches Operating in the Biological Window
K. Klaue, Y. Garmshausen, S. Hecht*
2017
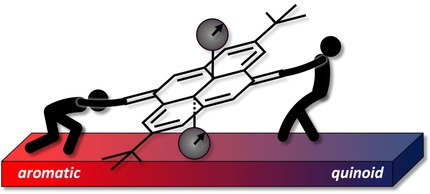
Dihydropyrene as an Aromaticity Probe for Partially Quinoid Push-Pull Systems
Y. Garmshausen, K. Klaue, S. Hecht*